Evaluation and diploma
Evaluation
Each Consortium member, will evaluate the students by continuous assessment and formal exams under the responsibility of:
- Prof. Andrieux, Prof. Alhareth and Prof. Yohann Corvis for Université Paris Cité
- Prof. Antimisiaris and Prof. Mourtas for Panepistimio Patron
- Prof. Caramella, Prof. Conti and Prof. Terreni for Universtà degli Studi di Pavia
- Prof. Roger, Prof. Venier-Julienne and Prof. Bastiat for Université d’Angers
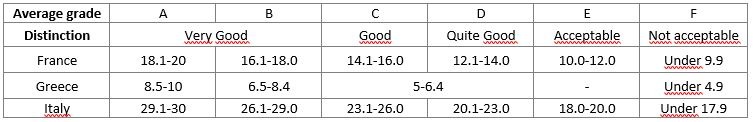
Students will be evaluated according to the following grading system:
To pass a study unit or a course, the student must obtain at least an E mark. At the end of each semester, the student must validate 30 ECTS. The corrected tests will be open to consultation after addressing a formal request to the professor in charge; all kinds of exams (written, oral presentation, individual report etc.) will be stored for one year after the end of the intake at the member of the Consortium in which the exam took place.
During Semester 4, all students do an internship in a private company or in a research laboratory collaborating with the Consortium. The internship must last 24 to 26 weeks. The location and the topic of the internship will have be validated by the Consortium beforehand. Its assessment will be based on the written report (40%) of the student, the evaluation of the local supervisor (20%) and the oral defense (40%). The defense of the Master thesis will be presented viva voce during the workshop following the summer school, in front of an International jury of Nanomedicine experts from Associated Partners, private companies and the NANOMED Consortium. The final mark will be delivered by the Consortium
Re-sits
In case of failure, a single re-sit examination will be organized; should the student fail again, he/she will have to quit the NANOMED Master.
In case of failure for the final dissertation, the student will have the opportunity to:
- Prepare a new oral presentation before the end of July of the graduating year;
- Prepare a new version of the report and a new oral presentation that will have to take place before the end of July of the graduating year;
- Attend a new traineeship and renew the whole process, provided that the supervising Consortium member agrees to it.
However, if the final exam (written report and oral presentation) cannot happen before the end of September of the graduating year, the student must register again for the following academic year at his/her own expenses and in each university of the Consortium, regardless of his/her previous status of scholarship holder or self-funded student. This new registration is mandatory for the student to validate his/her work and thus to complete the NANOMED programme and be allowed to graduate. The second final exam will be held after the second internship is completed. The Executive committee will communicate to the student the deadline for sending the written report and will set a date for the oral presentation at his/her convenience.
Graduation
To obtain the final diplomas (one from each Consortium member) and the diploma supplement, students must validate 4 semesters. The degrees will be delivered according to the decision of the Executive committee composed of at least one representative per Consortium member.
À lire aussi

SAVE THE DATE: Workshop 2025
We are pleased to announce that the 8th NANOMED Workshop will be organized by the Faculty of Pharmacy, Université Paris Cité ! It will take place on 8, 9, 10 and 11 July 2025. This edition of the workshop will focus on "Green Nanomedicine and Advanced Biotherapy",...

FOCUS: Students’ work
Congratulations to our Nanomed students for their recent publications! This month, we are extremely happy to announce that one of our students has seen his work accepted for publication. 1. Co-encapsulation of fisetin and cisplatin into liposomes: Stability...

Invited Professor: Mr Dimitrios Lamprou
The NANOMED EMJMD was delighted to welcome Prof. Dimitrios Lamprou, from Queen's University Belfast, for a 2-week course in November 2023 at the University of Pavia! Prof. Dimitrios Lamprou (Ph.D. MBA) is the Chair of Biofabrication and Advanced Manufacturing &...

APGI Info Day in Paris
The students from our seventh intake participated in the Info Day “Poorly soluble drugs: An industrial perspective on formulations for bio-availability enhancement” organized by APGI (“Association de Pharmacie Galénique Industrielle” or “International Society for Drug...